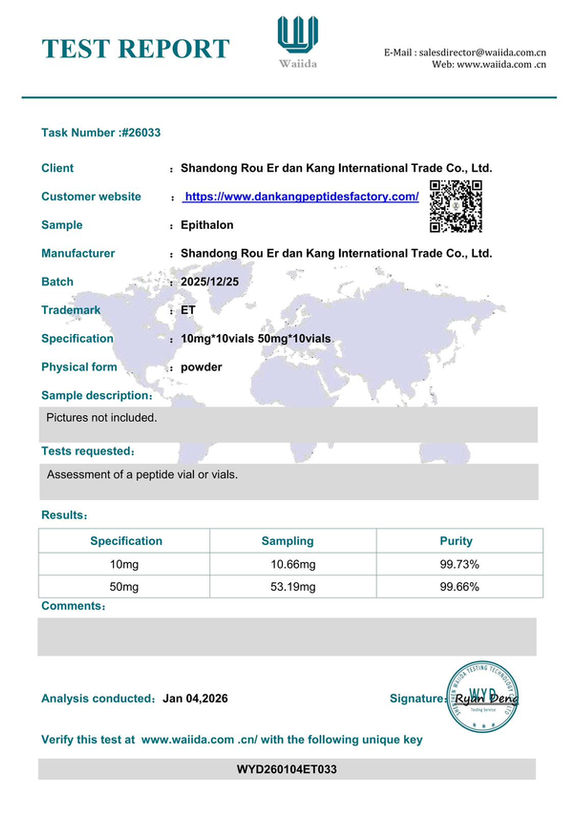
Lab Testing & Purity Reports
Every batch is independently tested by Waiida Testing Technology. View our certificates and verify results directly.
COA
Certificate
of
Authenticity
Why is this so important?
Quality & Safety
-
Peptides are research compounds with high purity requirements. A COA documents the exact purity level (typically 95%+, sometimes 99%+), which directly affects research validity and safety.
-
It confirms the product hasn't been contaminated, degraded, or substituted with cheaper/harmful alternatives.
Research Integrity
-
If you're using peptides for research, a COA proves your baseline quality. Results are only reproducible if the input material is verified.
-
Peer review, regulatory bodies, and collaborators often require documented purity/authenticity to validate findings.
Regulatory & Legal Protection
-
Peptides exist in a gray zone (research vs. therapeutic use). A COA from a reputable supplier creates a paper trail showing you sourced from a legitimate vendor—important if regulatory questions arise.
-
Protects you if the product is later questioned or recalled.
Supplier Credibility
-
A vendor willing to issue detailed COAs (with batch numbers, testing dates, third-party lab verification) signals they stand behind their product and have quality controls in place.
-
Vendors without COAs are often red flags for lower quality or unverified sourcing.
What a strong peptide COA should include:
-
Peptide name & batch/lot number
-
Purity percentage (e.g., "98.5% HPLC verified")
-
Molecular weight confirmation
-
Testing date & expiration/stability info
-
Third-party lab name (not just the supplier's own lab)
-
Certificate number for traceability